CONTRAINDICATIONS
Hypocalcemia: Pre-existing hypocalcemia must be corrected prior to initiating therapy with AUKELSO.
Hypersensitivity: AUKELSO is contraindicated in patients with known clinically significant hypersensitivity to denosumab products.
WARNINGS AND PRECAUTIONS
Drug Products with Same Active Ingredient
Patients receiving AUKELSO should not receive other denosumab products concomitantly.
Hypersensitivity
Clinically significant hypersensitivity including anaphylaxis has been reported with use of denosumab products. Reactions may include hypotension, dyspnea, upper airway edema, lip swelling, rash, pruritus, and urticaria. If an anaphylactic or other clinically significant allergic reaction occurs, initiate appropriate therapy and discontinue AUKELSO therapy permanently.
Hypocalcemia
Denosumab products can cause severe symptomatic hypocalcemia, and fatal cases have been reported. Correct pre-existing hypocalcemia prior to AUKELSO treatment. Monitor calcium levels, throughout AUKELSO therapy, especially in the first weeks of initiating therapy, and administer calcium, magnesium, and vitamin D as necessary. Concomitant use of calcimimetics and other drugs that can lower calcium levels may worsen hypocalcemia risk and serum calcium should be closely monitored. Advise patients to contact a healthcare provider for symptoms of hypocalcemia.
An increased risk of hypocalcemia has been observed in clinical trials of patients with increasing renal dysfunction, most commonly with severe dysfunction (creatinine clearance less than 30 mL/min and/or on dialysis), and with inadequate/no calcium supplementation. Monitor calcium levels and calcium and vitamin D intake.
Osteonecrosis of the Jaw (ONJ)
ONJ has been reported in patients receiving denosumab products, manifesting as jaw pain, osteomyelitis, osteitis, bone erosion, tooth or periodontal infection, toothache, gingival ulceration, or gingival erosion. Persistent pain or slow healing of the mouth or jaw after dental surgery may also be manifestations of ONJ. In clinical trials in patients with cancer, the incidence of ONJ was higher with longer duration of exposure. Risk factors include a history of tooth extraction, poor oral hygiene, or use of a dental appliance. Other risk factors include immunosuppressive therapy, treatment with angiogenesis inhibitors, systemic corticosteroids, diabetes, and gingival infections, and a history of invasive dental procedures for denosumab-treated patients with multiple myeloma.
Perform an oral examination and appropriate preventive dentistry prior to the initiation of AUKELSO and periodically during AUKELSO therapy. Advise patients regarding oral hygiene practices. Avoid invasive dental procedures during treatment with AUKELSO. Consider temporary discontinuation of AUKELSO therapy if an invasive dental procedure must be performed.
Patients who are suspected of having or who develop ONJ while on AUKELSO should receive care by a dentist or an oral surgeon. In these patients, extensive dental surgery may exacerbate the condition.
Atypical Subtrochanteric and Diaphyseal Femoral Fracture
Atypical femoral fracture has been reported with denosumab products. These fractures can occur anywhere in the femoral shaft from just below the lesser trochanter to above the supracondylar flare and are transverse or short oblique in orientation without evidence of comminution.
Atypical femoral fractures most commonly occur with minimal or no trauma to the affected area. They may be bilateral and many patients report prodromal pain in the affected area, usually presenting as dull, aching thigh pain, weeks to months before a complete fracture occurs. A number of reports note that patients were also receiving treatment with glucocorticoids (e.g., prednisone) at the time of fracture.
During AUKELSO treatment, patients should be advised to report new or unusual thigh, hip, or groin pain. Any patient who presents with thigh or groin pain should be evaluated to rule out an incomplete femur fracture. Patient presenting with an atypical femur fracture should also be assessed for symptoms and signs of fracture in the contralateral limb. Interruption of AUKELSO therapy should be considered, pending a risk/benefit assessment, on an individual basis.
Hypercalcemia Following Treatment Discontinuation in Patients with Giant Cell Tumor of Bone (GCTB) and in Patients with Growing Skeletons
Clinically significant hypercalcemia requiring hospitalization and complicated by acute renal injury has been reported in denosumab product-treated patients with GCTB and patients with growing skeletons within the first year after treatment discontinuation. After treatment is discontinued, monitor patients for signs and symptoms of hypercalcemia and manage patients as clinically appropriate.
Multiple Vertebral Fractures (MVF) Following Treatment Discontinuation
MVF have been reported following discontinuation of treatment with denosumab products. Patients at higher risk for MVF include those with risk factors for or a history of osteoporosis or prior fractures. When AUKELSO treatment is discontinued, evaluate the individual patient’s risk for vertebral fractures.
Embryo-Fetal Toxicity
Based on data from animal studies and its mechanism of action, denosumab products can cause fetal harm when administered to a pregnant woman.
Advise females of reproductive potential to use effective contraception during therapy and for at least 5 months after the last dose of AUKELSO. Advise pregnant women and females of reproductive potential that exposure to AUKELSO during pregnancy or within 5 months prior to conception can result in fetal harm.
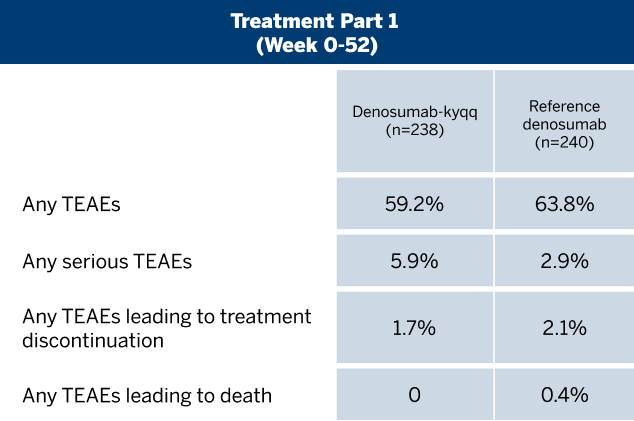
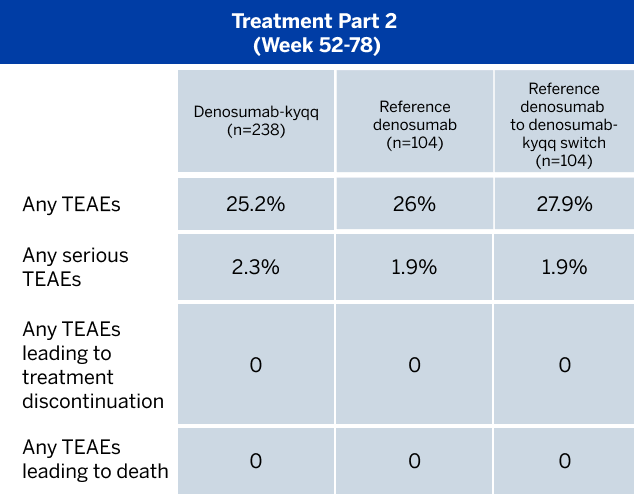
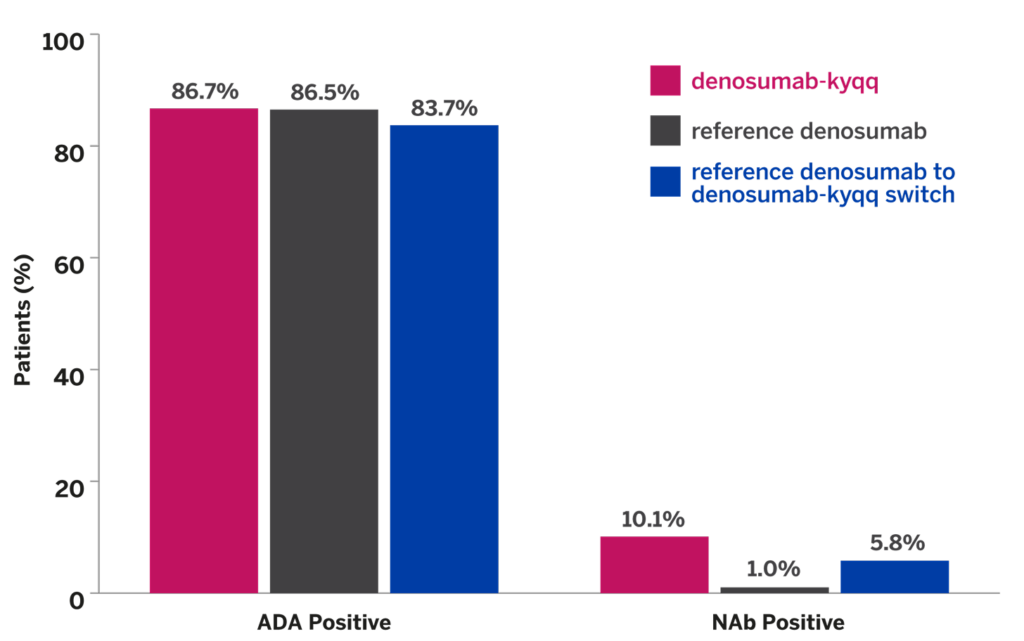
ADVERSE REACTIONS
The most common adverse reactions (incidence ≥25%) in patients receiving denosumab with bone metastasis from solid tumors were fatigue/asthenia, hypophosphatemia, and nausea. The most common serious adverse reaction was dyspnea. The most common adverse reactions resulting in discontinuation of denosumab were osteonecrosis and hypocalcemia.
The most common adverse reactions in patients receiving denosumab with multiple myeloma (incidence ≥10%) were diarrhea, nausea, anemia, back pain, thrombocytopenia, peripheral edema, hypocalcemia, upper respiratory tract infection, rash, and headache. The most common serious adverse reaction (incidence ≥5%) was pneumonia. The most common adverse reaction resulting in discontinuation of denosumab (≥1.0%) was osteonecrosis of the jaw.
The most common adverse reactions in patients receiving denosumab with giant cell tumor of bone (incidence ≥10%) were arthralgia, back pain, pain in extremity, fatigue, headache, nausea, nasopharyngitis, musculoskeletal pain, toothache, vomiting, hypophosphatemia, constipation, diarrhea, and cough. The most frequent serious adverse reactions were osteonecrosis of the jaw (3.6%), bone giant cell tumor (1.5%), anemia (1.1%), pneumonia (0.9%), and back pain (0.9%). The most frequent adverse reactions resulting in discontinuation of denosumab was osteonecrosis of the jaw (incidence of 3.6%). The adverse reaction profile appeared similar in skeletally mature adolescents and adults.
Adverse reactions occurring in >20% of patients receiving denosumab with hypercalcemia of malignancy were nausea, dyspnea, decreased appetite, headache, peripheral edema, vomiting, anemia, constipation, and diarrhea. The following adverse reactions of Grade 3 or greater severity related to study therapy were reported on-study: fatigue (3%) and infection (6%). Grade 3 laboratory abnormalities included hypomagnesemia (3%), hypokalemia (3%), and hypophosphatemia (76%) of patients. No deaths on-study were related to denosumab therapy.